ISO 13485 Medical Devices Quality Management System (MDQMS) - Training Courses
Foundation Training
ISO 13485 Medical Devices Quality Management System (MDQMS) - Training Courses
SUMMARY
ISO 13485 Foundation training enables you to learn the basic elements to implement and manage a Medical Devices Quality Management System (MDQMS) as specified in ISO 13485. During this training course, you will be able to understand the different modules of a MDQMS, including MDQMS policy, procedures, performance measurements, management commitment, internal audit, management review and continual improvement.
After completing this course, you can sit for the exam and apply for the "PECB Certified ISO 13485 Foundation" credential. A PECB Foundation Certificate shows that you have understood the fundamental methodologies, requirements, framework and management approach.
Who Should Attend?
- Individuals involved in Medical Devices Quality Management
- Individuals seeking to gain knowledge about the main processes of Medical Devices Quality Management Systems (MDQMS)
- Individuals interested to pursue a career in Medical Devices Quality Management
COURSE AGENDA
DURATION: 2 Days
Day 1
Introduction to Medical Devices Quality Management System (MDQMS) concepts as required by ISO 13485
Day 2
Medical Devices Quality Management System requirements and Certification Exam
Learning Objectives
- Understand the elements and operations of a Medical Devices Quality Management System (MDQMS)
- Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks
- Understand the approaches, methods and techniques used for the implementation and management of a MDQMS
EXAMINATION & CERTIFICATION
The PECB certified ISO 13485 Foundation Training Course exam fully meets the requirements of the PECB Examination and Certification Program (ECP). The exam covers the following competency domains:
1
Domain 1: Fundamental principles and concepts of a Medical Devices Quality Management System (MDQMS)
2
Domain 2: Medical Devices Quality Management System (MDQMS)
CERTIFICATION
After successfully passing the exam, you can apply for the credential shown on the table below. You will receive the certificate once you comply with all the requirements related to the selected credential. Certification fees are included in the exam price.
Credentials
PECB Certified ISO 13485 Foundation
Exam
PECB Certified ISO 13485 Foundation exam or equivalen
Professional Experience
None
MS Audit / Assessment Experience
None
MDQMS Project Experience
None
Other Requirements
Signing the PECB Code of Ethics
GENERAL INFORMATION
- Certification fees are included on the exam price.
- Training material containing over 200 pages of information and practical examples will be distributed.
- A participation certificate of 14 CPD (Continuing Professional Development) credits will be issued.
- In case of exam failure, you can retake the exam within 12 months free of charge.
Training Calendar
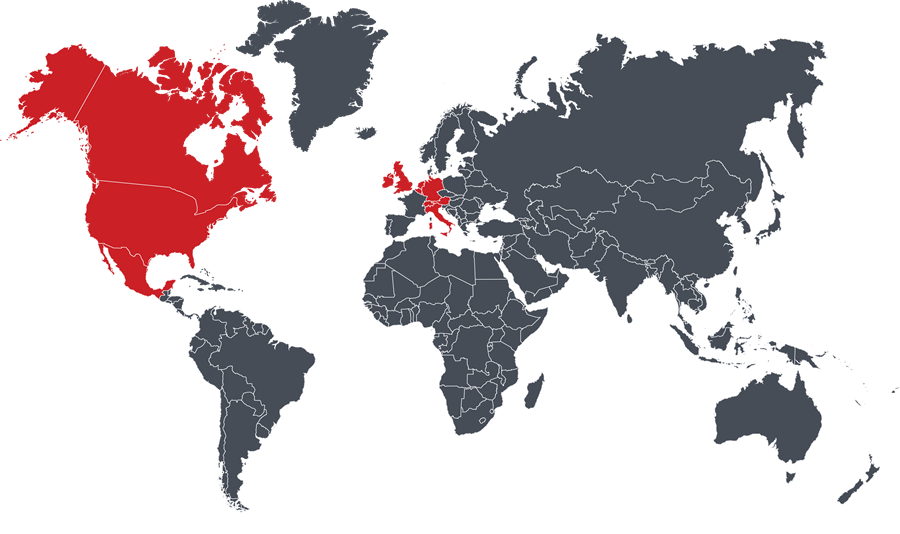
Austria, Belgium, Canada, Germany, Ireland, Italy, Lichtenstein, Luxembourg, The Netherlands, Switzerland, United Kingdom, United States
|
Course |
Dates |
Location |
Cost |
Pay Now |
Request Seat |
| Self Study Course |
Online |
$ 710 |
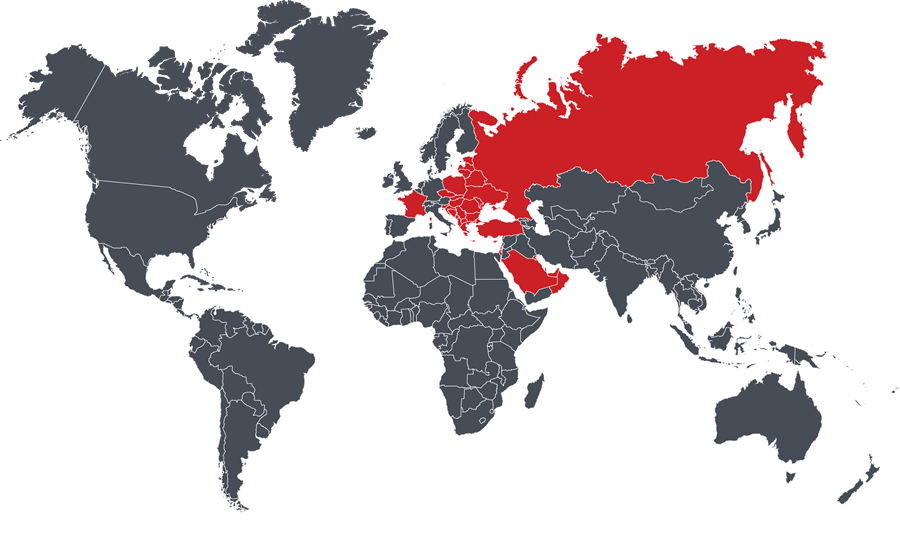
Albania, Bahrain, Belarus, Bosnia & Herzegovina, Bulgaria, Croatia, Czech Republic, Cyprus, Estonia, France, Greece, Hungary, Israel, Kosova, Kuwait, Latvia, Lithuania, Macedonia, Malta, Moldova, Montenegro, Oman, Poland, Qatar, Romania, Russia, Saudia Arabia, Serbia, Slovakia, Slovenia, Turkey, Ukraine, United Erab Emirates
|
Course |
Dates |
Location |
Cost |
Pay Now |
Request Seat |
| Self Study Course |
Online |
$ 510 |
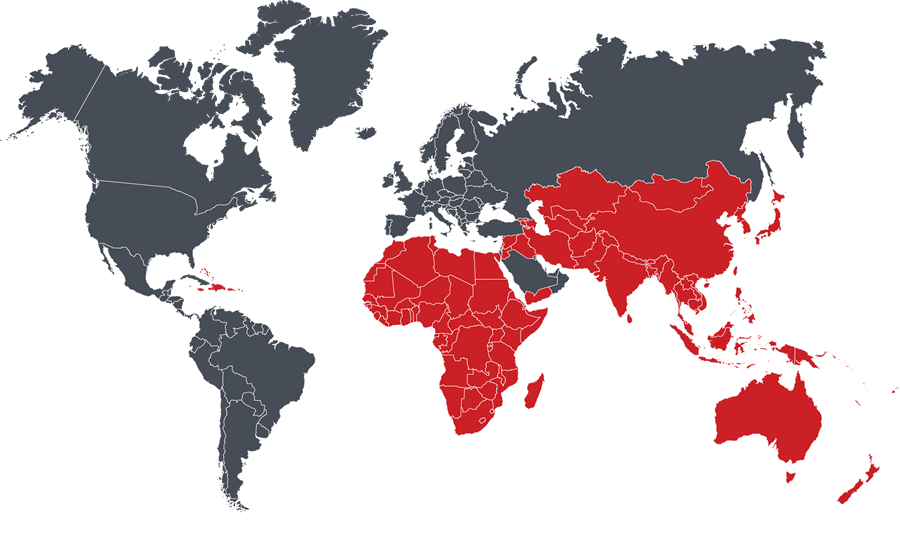
Afghanistan, Algeria, American Samoa, Angola, Antigua & Barbuda, Armenia, Australia, Azerbaijan, Bahamas, Bangladesh, Barbados, Benin, Bhutan, Botswana, Brunei, Burkina Faso, Burundi, Cambodia, Cameroon, Cape Verde, Central African Republic, Chad, China, Comoros, Cook Islands, Ivory Coast, Dominica, DR Congo, Djibouti, East Timor, Egypt, Equatorial Guinea, Eritrea, Ethiopia, Fiji, Gabon, Georgia, Ghana, Grenada, Guam, Guinea, Guinea-Bissau, Haiti, Hong Kong, India, Indonesia, Iran, Iraq, Jamaica, Japan, Jordan, Kazakhstan, Kenya, Kiribati, Kyrgyzstan, Laos, Lebanon, Lesotho, Liberia, Libya, Madagascar, Macau, Malawi, Malaysia, Maldives, Mali, Marshall Islands, Mauritania, Mauritius, Mayotte, Micronesia, Mongolia, Morocco, Mozambique, Myanmar, Namibia, Nauru, Nepal, New Caledonia, New Zealand, Niger, Nigeria, Niue, Norfolk Islands, North Korea, North Mariana Island, Pakistan, Palau, Palestine, Papua New Guinea, Philippines, Pitcaim Islands, Puerto Rico, Republic of the Congo, Rwanda, Saint Helena, Saint Kitts & Nevis, Saint Lucia, Saint Vincent & Grenadines, Samoa, Sao Tome and Principe, Senegal, Seychelles, Sierra Leone, Singapore, Solomon Islands, Somalia, South Africa, South Korea, South Sudan, Sri Lanka, Sudan, Swaziland, Syria, Tajikistan, Taiwan, Tanzania, Thailand, The Gambia, Togo, Tonga, Trinadad and Tobago, Tunisia, Turkmenistan, Tuvalu, Uganda, US Virgin Islands, Uzbekistan, Vanuatu, Vietnam, Wallis and Futuna, Western Sahara, Yemen, Zambia, Zimbabwe
|
Course |
Dates |
Location |
Cost |
Pay Now |
Request Seat |
| Self Study Course |
Online |
$ 410 |
Would you like a Foundation Training in your area?


